Is that teenage dose of glandular fever driving your MS?
New research has confirmed the link between Epstein-Barr (the glandular fever virus) and multiple sclerosis. But if nearly everyone carries this virus, why do so few people develop the disease? Is targeting EBV really the answer we've all been waiting for?
If you've been diagnosed with MS, or you're deep in that exhausting limbo of chasing answers and getting nowhere, there's a good chance you've come across something in recent months. A growing discussion or conversation thread that says: “Epstein-Barr virus caused my MS”.
I totally understand why you'd want to pull on that thread. Because at last, after years of being handed a diagnosis with very little explanation, being told by your consultant you were “just unlucky”, here is finally something…a cause, a name, a reason.
So I want to clear up the confusion with you today, because I think it matters enormously and here’s my view: EBV is almost certainly part of your story and in many cases, a significant part. What it's unlikely to be, though, is the whole story.
By the end of this post, you'll understand why EBV alone doesn't explain MS, what the research actually tells us and why that's not a dead end but rather something genuinely useful. You'll also understand why the question to ask isn't "How do I get rid of EBV?" but rather something far more helpful and far more actionable.
You've probably felt this
You've done the research. You've gone down the Google rabbit hole at 11pm when sleep is evading you. You've maybe tried antivirals, high-dose vitamin C, protocols you found on Facebook groups run by people who sound very convincing. But then, a few months later, you still feel the same, or worse.
This is what happens when one piece of a genuinely complex puzzle is treated as if it's the whole picture. The research on EBV and MS is real, current and important. The clinical oversimplification of it, though, is causing a lot of confusion and doing a lot of people harm. So let’s break it down.
What the research actually says
Humour me for a moment with some science geek-ery (I promise to make it simple).
EBV is a member of the pesky herpesvirus family, best known for causing glandular fever (infectious mononucleosis). Like all herpesviruses, it doesn't leave the body after the initial infection. So yes, that teenage bout of glandular fever is still in your body. I’m sure we’re all familiar with those frustrating cold sores (also herpes family) that rear their heads just before a key presentation or stressful event - that’s how herpes family viruses work, they stay with us foreverrrr, sigh. After the initial symptoms have gone, EBV goes to sleep (becomes latent), tucking itself comfortably inside our memory B cells, a type of immune cell that stores the body's immunological memory.
The research linking EBV to MS has become increasingly compelling. A recent US military study, following over 10 million service personnel over two decades, found that EBV infection increased the risk of subsequently developing MS by 32x. Prior to EBV infection, the risk of MS remained low; after it, the risk increased sharply. The study's authors concluded that “EBV is a necessary precursor to MS development in most cases”. [1]
OK, great, so that’s really important, but here is the number that changes evvverything.
A 2025 French study, analysing nearly 20,000 EBV samples, found that fewer than 3% of adults remain negative after the age of 25. Which effectively means that EBV prevalence in the adult population is, in practice, almost 97%... pretty much all of us! [2]
Around 150,000 people in the UK are estimated to be living with MS. Roughly 70-75% of them are women. Yet EBV is present in almost the entire adult population. So let’s do the simple maths, if EBV caused MS, we would expect the numbers to look very different indeed.
So the phrase that matters most in all of this is: EBV is necessary, but NOT sufficient for MS.
It's a required contributor, but it’s not the full explanation.
Donna's story
Donna came to me three years after her MS diagnosis, which had arrived in her late 40s following a bout of optic neuritis and an MRI showing lesions consistent with relapsing remitting disease. She'd had glandular fever at 14. Recovered, didn't think much of it… got on with life.
However, when we sat down and mapped her timeline properly, something more layered became apparent.
As a child, she'd had ear infections, tonsillitis, scarlet fever, German measles, multiple courses of antibiotics, then a tonsillectomy at eight years old. In her teens, alongside the glandular fever (EBV), she started the oral contraceptive pill and stayed on it for over twenty years. Her adult life was high-pressure and relentless: a senior role in financial services, frequent travel, irregular sleep, eating on the run, plus all the usual life stuff that high-performing women still have to deal with.
The subtle signals were there over the years. Fatigue that she pushed through. Periods of low resilience she brushed off as stress. Digestive symptoms that were all too common in friends. Nothing that, in isolation, would trigger alarm.
Then she hit perimenopause coupled with a nasty dose of Covid-19.
The hormonal shift and another nasty virus, seemingly acting together as a kind of tipping point. Neurological symptoms appeared and an eventual diagnosis, after lots of fighting and the standard initial dismissal of “it’s just the menopause” (please, don’t even get me started on women having to fight to get heard in the autoimmune world).
Was EBV part of Donna's story? Absolutely. Was it the cause of her MS? I would argue it was more like a key player in an already-loaded deck. By the time her immune system ran into trouble, the foundations had likely been under pressure for decades.
Why immune ‘competence’ is the real question
This change in thinking makes a huuuge difference.
Rather than asking “Is EBV present?” the more useful question is “Why is the immune system no longer able to keep EBV in check?”
Because, when someone is immunologically ‘competent’, when the immune system is well-nourished, well-regulated and not under excessive burden, EBV sits quietly in its sleepy dormant state. It doesn't cause much trouble. It simply waits, opportunistically…
The problem arises when the immune system's capacity is stretched. When nutrient deficiencies accumulate. When gut health deteriorates. When chronic stress drives sustained cortisol and depletes immune reserves. When toxin load climbs. When oestrogen, which has known neuroprotective and immunomodulatory effects, drops sharply as it does in perimenopause and menopause.
Under these conditions, latent EBV takes the opportunity and can reactivate; and in someone with the relevant genetic susceptibilities, with a history of early-life immune insults, with accumulated inflammatory burden, that reactivation can tip the immune system into the kind of dysregulation that, over time, leads to demyelination.
The virus doesn't cause the disease in isolation. The depleted immune foundations makes the disease possible.
The EBV–COVID connection: a warning signal hiding in plain sight
One of the most striking developments in recent years is what Covid-19 has revealed about EBV reactivation and autoimmune conditions in general.
In 2023 it was uncovered that EBV reactivated in 27.1% of Covid-positive patients compared to 12.5% in healthy controls. But the Long Covid data is where it gets really significant.[3]
Studies on Long Covid patients found EBV reactivation in 66.7% of cases, compared with just 10% of those recovered with no symptoms. Covid, it seems, is unusually effective at waking a virus that most immune systems had long since put to bed.[4]
So Covid-19 seems to act as a powerful immune stressor that reactivates latent EBV in a significant proportion of people. For those with an already-burdened immune system, that reactivation has downstream consequences: fatigue, brain fog, myalgia and in those with existing autoimmune predisposition, potentially a worsening or new onset of autoimmune activity.
The wave of new-onset autoimmunity we have seen post-pandemic may be partly explained by this mechanism. EBV reactivation doesn't occur in a vacuum; it occurs in a system under pressure.
(That relationship between Covid and autoimmunity is a whole conversation in itself. I'll be writing about it separately.)
What else is going on? The multiple drivers of MS
Once someone has MS, the picture becomes even more layered. EBV may have been a significant early trigger, but there are other immune variables that can perpetuate and amplify demyelination and not everyone with MS continues to have elevated EBV activity. This is important because if we only focus on EBV, we miss the variables that may currently be driving disease activity.
The key areas to look for:
Vitamin D status
Low vitamin D is strongly and consistently associated with both increased MS risk and increased disease activity. The UK is a difficult environment for vitamin D synthesis, especially across autumn and winter and deficiency is widespread. Optimising vitamin D is one of the highest-yield, lowest-risk interventions available (read more about the importance of vitamin D here).
Methylation and B vitamin status
Vitamins B2, B3, B12, B6 and folate, together with zinc, magnesium, choline and betaine are essential for methylation, the biochemical process that regulates gene expression, DNA repair, immune signalling and the management of viral latency (aka keeping EBV asleep!). Impaired methylation can compromise your immune competence over time and may affect how the body handles latent viral load.
Gut health
The gut microbiome has constant, bidirectional communication with the immune system, it’s constantly chatting. When there is an imbalance in the healthy gut bugs (dysbiosis), whether from antibiotics, diet, stress or prior infections, this can promote a more inflammatory immune environment. Short-chain fatty acids (SCFAs), produced when beneficial gut bacteria ferment dietary fibre, play a key role in regulating immune balance. They are consistently found to be reduced in people with MS.[5] Supporting the gut is not a ‘nice-to-have’ in MS, it's a core priority.
Hormonal transitions
Oestrogen has both neuroprotective and immunomodulatory effects, interacting directly with how EBV behaves inside B cells (possibly why women are 2-3x more likely to develop MS than men). It supports myelin maintenance and modulates inflammatory signalling. The swings in oestrogen during perimenopause and menopause can therefore destabilise immune balance in ways that increase inflammatory activity and this hormonal window appears to act as a ‘grand reveal’ (in not such a good way) for women who may have been carrying a pre-existing immune vulnerability for years.[6]
Circadian rhythm and sleep
The immune system runs on a daily rhythm. Regulatory T cells, natural killer cells, cytokine production, all of these follow circadian patterns. Chronic sleep disruption, irregular eating patterns and poor light exposure don't just affect energy, they also impact the immune system's capacity to regulate itself.
Blood sugar regulation
Fluctuating blood sugar drives an inflammatory signalling environment, increases oxidative stress and places sustained metabolic burden on cells, including neurons. In a nervous system already under immune attack, that additional metabolic load can just add fuel to the fire.
Omega-3 and inflammatory balance
The ratio between omega-3and omega-6 fatty acids has a significant bearing on the inflammatory environment. Modern diets can tend heavily towards omega-6 (found in processed foods and oils), driving a pro-inflammatory lipid profile. Correcting this ratio through food and targeted supplementation can meaningfully change the inflammatory background. One of my favourite clean supplement suppliers is Wiley’s Finest.
Food sensitivities
Certain dietary proteins like gluten and dairy, can interact with the immune system in MS and closely resemble proteins in the myelin sheath. When the immune system responds to gluten in a sensitised individual, it can simultaneously begin attacking myelin as collateral damage through a process called molecular mimicry. This is valid even outside of coeliac disease; non-coeliac gluten sensitivity alone has been shown to cause neurological problems including myelin damage [7]. Dairy follows a similar logic, with a recent study finding that antibodies to the main protein in cow's milk can cross-react and directly trigger demyelination. [8]
Other factors such as oxidative stress, environmental toxicities and mould can all add additional fuel to the immune system fire.
If EBV isn't the whole answer, what do you actually do?
The goal is not to “kill” EBV. That's not possible and the evidence that antiviral approaches alone alter MS outcomes remains very limited. The goal is to rebuild your immune foundations and competence, reduce the overall burden on the immune system and create an internal environment in which we keep EBV asleep and it has less reason and less opportunity to wake up and cause trouble.
That means:
Testing, not guessing. Knowing your vitamin D, B12, folate, omega-3 index, inflammatory markers and gut function gives you a map rather than a set of assumptions.
Addressing the gut as a priority. Supporting microbiome diversity and gut lining integrity through dietary fibre, fermented foods and, where indicated, targeted probiotics can shift the immune environment substantially.
Supporting methylation. For some women, particularly those with MTHFR and associated gene variants, this involves more targeted B vitamin support through diet and supplementation as needed. Genetic testing can help clarify this.
Managing the hormonal transition thoughtfully, in ways that support both immune function and neurological health.
Treating sleep and circadian rhythm as priorities, not lifestyle ‘nice to haves’.
Stabilising blood sugar, which reduces the inflammatory load and supports neurological energy metabolism.
Reducing dietary allergens that keep the immune system aggravated.
Oh and reducing stress…sorry, that one’s a non-negotiable for EBV. Chronic stress drives cortisol, which is one of the most reliable triggers for EBV reactivation.
These all work to shore up your foundations, and support the immune system's ability to do its job.
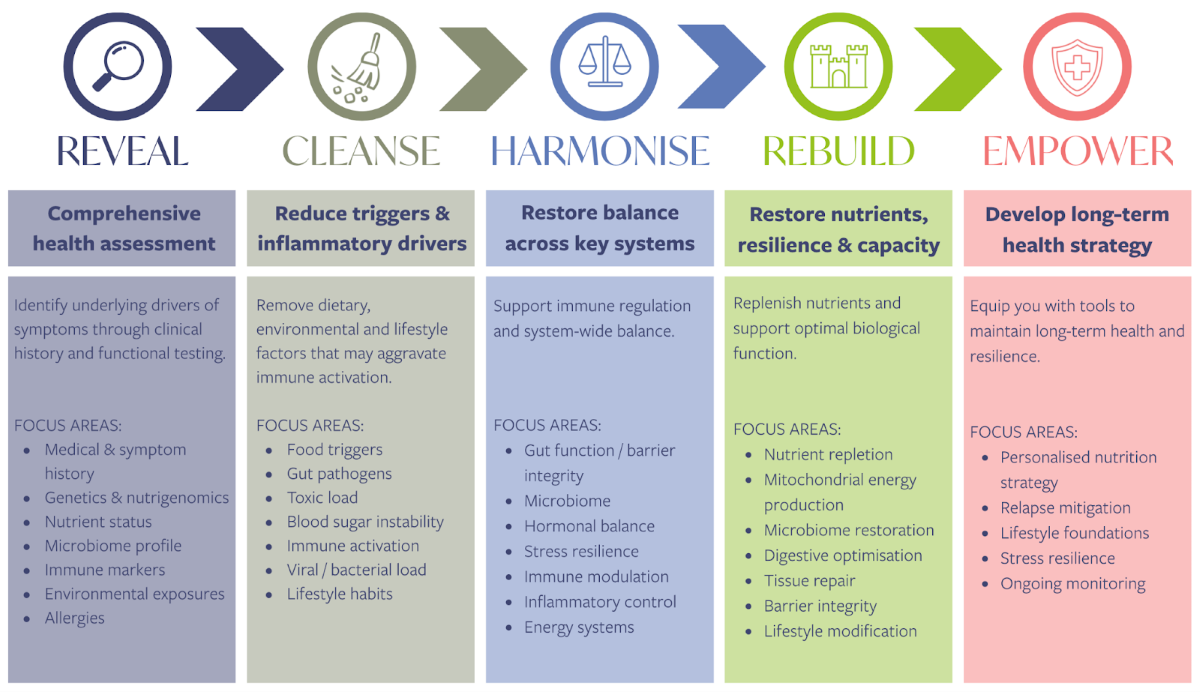
Working through all of this is, of course, where the individual picture really matters. My Get Empowered framework I use with clients follows five stages: Reveal, Cleanse, Harmonise, Rebuild and Empower. It sounds straightforward, but in practice each stage looks quite different for each person. For someone with MS, the reveal stage alone, properly mapping genetics, nutrient status, gut function, immune markers, environmental exposures, food allergens and viral history, often highlights things that may never have been on anyone's radar. The subsequent stages build on that, reducing the drivers we've uncovered, restoring balance across the systems that have been under strain and rebuilding the foundations that improve long-term resilience.
What I find consistently, is that the people who come to me have usually addressed one or two of these areas diligently, what’s often missing is the bigger picture. But as we work through the full map in a bespoke, layered and specifically sequenced way for your individual case, the picture becomes clear.
What next?
If you're living with MS, navigating a recent diagnosis or watching your symptoms change after a recent infection or Covid and wondering what on earth is going on, this is exactly the kind of complexity I work with and what I created my Get Empowered Programme for.
You don't have to keep piecing this together alone, after midnight, from forums and conflicting articles. If this feels like the right time to dig deeper into your own picture, I'd love to help you map it properly.
👉 Click here to book a free chat and let's talk about where you are and what might actually help. If you have a friend navigating MS, autoimmunity or Long Covid, please share this with them. Sometimes simply being shown the bigger picture can shift everything.
Skipped to the bottom? In summary
EBV and MS: the research is real, but the picture is bigger than one virus.
EBV is found in approximately 97% of adults. MS affects around 150,000 people in the UK, with 70-75% being women. The virus is clearly not acting alone.
EBV is likely a necessary contributor to MS in most cases, but it is not sufficient on its own. Genetic susceptibility, immune burden, gut health, nutrient status, hormone levels and chronic stress all shape whether the immune system can keep EBV dormant.
EBV reactivation is a significant feature of long Covid, occurring in 66.7% of long Covid patients versus 10% of controls. This may explain part of the post-pandemic rise in autoimmunity.[4]
The key question is not whether EBV is present, but why the immune system can no longer regulate it.
Supporting immune foundations through targeted testing, gut health, optimising vitamin status, blood sugar regulation, managing stress and sleep is likely to be more impactful than focusing solely on EBV.
References
1. Bjornevik, K. et al. (2022) ‘Longitudinal Analysis Reveals High Prevalence of Epstein-Barr Virus Associated with Multiple Sclerosis’, Science
2. Bernet, C. et al. (2025) ‘Evolution of Epstein-Barr Virus Infection Seroprevalence in a French University Hospital over 11 years, Including the COVID-19 Pandemic, 2013–2023’, Microorganism
3. Bernal, K.D.E. & Whitehurst, C. (2023) ‘Incidence of Epstein-Barr Virus Reactivation is Elevated in COVID-19 Patients’, Virology
4. Gold, J.E. et al. (2021) ‘Investigation of long COVID prevalence and its relationship to Epstein-Barr virus reactivation’, Pathogens
5. Tankou, S.K. et al. (2018) ‘A Probiotic Modulates the Microbiome and Immunity in Multiple Sclerosis’, Annals of Neurology
6. Keane, J.T. et al. (2021) ‘Gender and the Sex Hormone Estradiol Affect Multiple Sclerosis Risk Gene Expression in Epstein-Barr Virus-infected B Cells’, Frontiers in Immunology
7. Hadjivassiliou, M. Grünewald, R.A. & Davies-Jones, G.A.B. (2002) ‘Gluten Sensitivity as a Neurological Illness’, Journal of Neurology, Neurosurgery & Psychiatry
8. Chunder, R. et al. (2022) ‘Antibody Cross-reactivity between Casein and Myelin-associated Glycoprotein results in Central Nervous System Demyelination’, Proceedings of the National Academy of Sciences