Why your immune system isn't broken… it's confused (and what confused it in the first place)
The biology of autoimmunity explained in a way that actually makes sense
If you've recently been diagnosed with an autoimmune condition, or you've been unwell for a long time and suspect something immune-related may be going on, this post is for you.
By the time you reach the end, you'll understand what autoimmunity actually is, why your immune system is doing what it's doing and why that's not the whole story. We’ll dip into what was likely happening in your body long before you got a diagnosis and what that means for what's possible going forward.
This is the context nobody gives you in a 10-minute appointment. This is the explanation I needed and never got, so I wrote this to try and change that for you. Let’s dive in…
You're probably not confused about your symptoms. You're confused about the explanation
You've likely been living with unexplained symptoms like fatigue, joint pain, brain fog, digestive chaos, skin flare-ups or a combination of all of the above for longer than you care to remember. Then one day, after a string of appointments (sometimes across years), repeated blood tests and possibly a few dismissals along the way, someone handed you a diagnosis. Hashimoto's. Rheumatoid arthritis. Lupus. Multiple sclerosis. Coeliac disease. Ulcerative colitis. The list goes on…
What probably followed was a fairly brief explanation, maybe a leaflet (my personal experience), a prescription and a vague implication that this is now ‘just how things are’. That your immune system is ‘misfiring’, that there's not much to be done about it and that managing symptoms is now your new full-time job (as if you needed another one!).
Maybe they said “your body is attacking itself” or “your immune system has gone awry” and left it at that. Which, honestly, isn't the full story and not much to go on.
But before we go any further, I want you to understand that you are now part of a big group. A recent UK-wide study of 22 million people published in The Lancet found that autoimmune disorders now affect around 1 in 10 people overall, with women significantly overrepresented at 13% compared to 7% of men.[1] You are not unusual. You are not unlucky in some uniquely personal way. Your body has not failed you. You are part of a very large group of people whose immune systems are responding to a world and food environment that has changed far faster than our biology has been able to adapt to.
But crucially, the story is not over at the point of diagnosis. Not by a long way…
My story (why explanation matters)
In my early twenties I was working a high-pressure city job, pulling all-nighters, running on takeaways and stress and firmly believing I was invincible. Then a much-needed escape to an exotic beach came with an unexpected souvenir…a nasty gut bug that kicked off an autoimmune cascade in my body. Within days I was in agonising joint pain, unable to walk. My then-boyfriend (now husband) had to carry me around the island.
Once back in the UK, the search for answers began. That search took a looong time. Eventually, I was diagnosed with ankylosing spondylitis, an autoimmune inflammatory condition that can cause the vertebrae of the spine to fuse.
I vividly remember the consultant shaking my hand, saying “I'm sorry, there's nothing you can do”, and packing me off with a prescription for high-dose anti-inflammatories and a suggestion of future surgery. There was a leaflet, there was a feeble “I’m sorry”. There was, notably, very little else. No explanation of what had actually happened in my body, no discussion of the conditions that had allowed it to develop (no one ever asked about my history or the nasty gut bug), no map of the levers I might pull to influence what happened next. Just a handshake and a door.
What I needed, and didn't get, was the explanation I'm about to give you.
What followed was years of my own dogged research, driven by a refusal to believe that nothing could be done. I read everything I could find, slowly piecing together the picture of what had happened in my body and why. The occasional flare-up, the co-existing conditions that appeared over time, the frustrating inconsistency of symptoms, all of it gradually started to make sense. The real turning point came during a bout of uveitis when a more enlightened clinician at Moorfields (I wish I could remember his name as I am forever grateful), flagged that diet and internal immune terrain was legitimately part of the picture. That observation changed everything for me. Eventually I retrained and built my practice around exactly this kind of work. But that's another story.
The point is, explanation is not a luxury. It is the foundation of everything else. Once you understand what your immune system is doing and why, the whole picture changes and you have knowledge, power and opportunity for change.
What your immune system is actually trying to do
What your immune system is actually trying to do
Before you can understand what goes wrong in autoimmunity, you need to understand what the immune system is designed to do when it's working well.
Think of it as the world's most sophisticated border control operation, running 24 hours a day, 7 days a week, without a tea break. Its job is to check every molecule that crosses its path and make a decision: does this belong here… or doesn't it?
At its core, the immune system is a pattern recognition system.
More specifically it has to distinguish between self (the proteins and tissues that belong to you) and non-self (anything foreign that has entered the body). Within the non-self category, it has to make a further distinction: is this a friendly stranger (such as food proteins, pollen or harmless environmental bacteria), or a dangerous stranger (such as a pathogen that poses a genuine threat)?
When it identifies something as ‘dangerous’, it mounts an inflammatory response. That inflammation is not an error, it's the mechanism that keeps us alive. It's how the body removes threats and begins the process of repair - like, for example, when you get a virus. A healthy immune system is reactive, precise and, crucially, restorative. It fires up when you need it and stands down once the threat has been dealt with.
Tolerance is the word used to describe the immune system's ability to remain calm in the presence of things that are not a threat. Tolerance to your own tissues. Tolerance to food proteins. Tolerance to the trillions of friendly bacteria that live in your gut and are fundamental to your health. When that tolerance is maintained, things run smoothly, but when it starts to break down, the conditions for autoimmunity are set.
So what is autoimmunity, really?
Autoimmunity is what happens when immune tolerance to the body's own proteins is lost. The immune system, instead of recognising your own tissues as self, begins to treat them as foreign.
To understand how it does this, let’s briefly look at how antibodies work. An antibody is a protein produced by the immune system, shaped precisely to latch onto one specific target (called an antigen), a bit like a ‘lock-and-key’. When the immune system encounters something it has flagged as a threat, it produces antibodies that bind to it, marking it for destruction. Normally, this process is beautifully precise, something to be genuinely marvelled at and is how we battle viruses. In autoimmunity, those same antibodies start targeting the body's own tissues.
These are what we call ‘autoantibodies’ and their presence is usually what shows up in the blood tests that eventually lead to a diagnosis. But once autoantibodies bind to self-tissue, they don't just sit there. They can trigger inflammation directly, or they can form what are known as ‘immune complexes’, clusters of antibodies, antigens and immune cells that accumulate and deposit in tissues. Imagine your immune system has sent in the clean-up crew, but instead of leaving once the job is done, they set up camp and make a bit of a mess. In conditions like lupus and rheumatoid arthritis, these complexes can deposit in joints, kidneys and blood vessel walls, driving ongoing inflammation and damage even when the original trigger is long gone.[2] It is one of the reasons why autoimmune conditions can feel so persistent and so unpredictable because the immune response has effectively taken on a life of its own.
Over time, if that attack continues unchecked, it damages the tissue in question. In Hashimoto's, that tissue is the thyroid gland. In rheumatoid arthritis, it's the joints. In multiple sclerosis, it's the myelin sheath around the nerves. In coeliac disease, it's the lining of the small intestine. The diagnosis you receive is essentially telling you where the attack is focused, but it says very little about why the attack started in the first place.
Here's the part that might surprise you and that I think is important to emphasise.
The immune system is not making a mistake. It is doing exactly what it is designed to do…identifying a protein it has been primed to see as foreign and attacking it.
So WHY on earth, and HOW, does the immune system get primed against our own body’s tissues??
The case of mistaken identity
Molecular mimicry is one of the leading mechanisms by which infections or chemical agents can trigger autoimmunity. It occurs when the structural similarities between a foreign protein (from a virus, bacteria or other agent) and the body's own proteins are close enough that the immune system, in mounting its attack on the invader, ends up targeting self-tissue too.[3]
Picture a wanted poster going up in your immune system after a viral infection. The poster says:
“Look for anything that resembles this protein!”
Your immune cells memorise the description, get to work and successfully clear the threat. Job done. Except that, in some cases, the description on the poster looks uncannily like one of your own body's proteins, say, thyroid tissue for example. The immune system, doing precisely what it was told, keeps scanning, and keeps finding matches in your thyroid tissue and tagging it for destruction. It is not misbehaving, it’s been genuinely misled.
This is important, because it opens up a very different set of questions about what to do next. You can read more about the three conditions that come together for autoimmunity to develop in my post on the autoimmune triad.
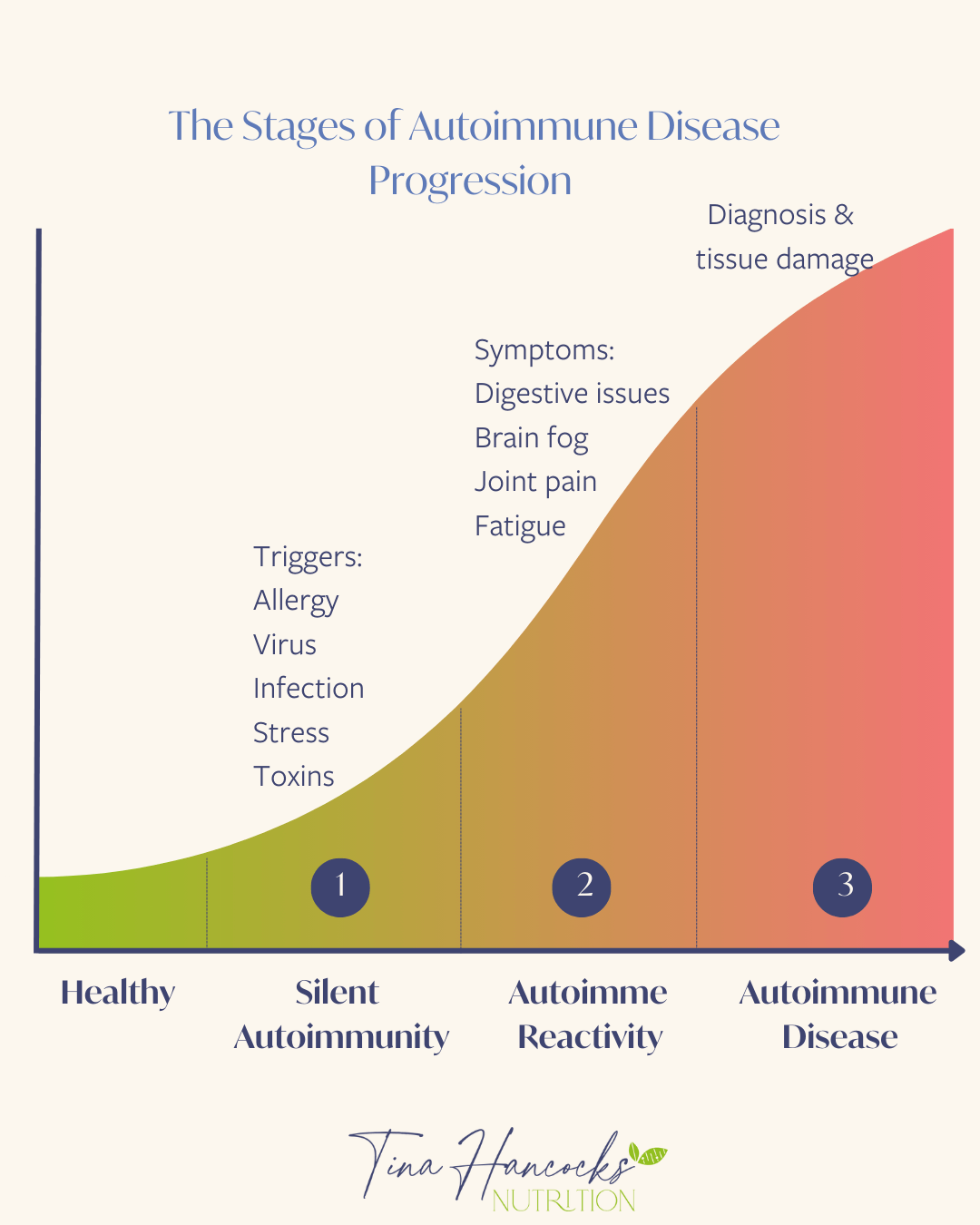
The long runway you didn't know you were on
One of the most significant and underappreciated facts about autoimmune disease is how long it typically develops before a diagnosis is made.
Autoantibody levels can rise years before any autoimmune symptoms or clinical diagnosis appears. Research into lupus, for example, has found that autoantibodies against nuclear antigens were detected an average of 5.6 years before the onset of symptoms and 8.7 years before diagnosis in individuals who later developed the condition.[4] Similar findings exist across rheumatoid arthritis, systemic sclerosis, type 1 diabetes and other autoimmune conditions.[5]
What this tells us is that autoimmune disease does not arrive out of nowhere.
There is a germination period, often a long one, during which the immune system is changing, reacting, accumulating dysregulation. By the time symptoms become undeniable and a diagnosis follows, the process has typically been underway for years. Long, possibly, before you connected your fatigue with your stress levels, or your gut symptoms with your joint pain. You can read more about the continuum of the autoimmune spectrum here
This is not meant to be alarming, but to help clarify things. Because it means that what you are dealing with is not a sudden malfunction that came on after your Saturday night out, but an evolving pattern and patterns can be understood, tracked and, with the right approach, influenced.
Hey there - short interlude…
If this is making you realise your body is craving more space, stillness and time outdoors, you are not alone.
Living with autoimmune disease can ask a lot of the body and mind, navigating fatigue, pain and flare-ups. While nature is not a cure, creating space to rest, breathe, move gently and feel held by the natural world can be a deeply nourishing and supportive part of your wellbeing.
This is exactly why I created my upcoming Nourished by Nature retreat day.
Now, back to the article...
Why the same diagnosis looks so different from person to person
What I often see is a confusion in comparing notes with others who share your diagnosis and finding that your experience looks completely different.
One person with Hashimoto's may have crippling fatigue and weight gain. Another may have anxiety and insomnia. One person with rheumatoid arthritis responds well to a particular protocol or medication. Another does not. Same diagnosis, completely different presentations, completely different responses to intervention.
This isn’t random or an anomaly, it reflects a fundamental truth about autoimmune and autoinflammatory disease - they are not fixed, distinct entities. They exist on a continuum. The inflammatory process that drives them is shaped by the particular combination of genetic predisposition, environmental exposures, gut / lung / skin barrier integrity, microbiome composition, stress history, hormonal context and nutritional status that is unique to each person.
The same trigger can lead to vastly different outcomes depending on the body (and the internal terrain) that it lands in.
This is why a purely diagnosis-led treatment approach, essentially assigning a condition and a matching medication, has its limits. The diagnosis tells you what is being attacked. It doesn't tell you what created the conditions for the attack, or what is keeping it going.
What actually influences immune tolerance?
Immune tolerance is not a fixed setting. It is an active, dynamic process that is influenced by a number of factors that sit, for the most part, well within the territory of nutritional and functional medicine.
The gut barrier plays a central role. The gut lining is ideally a selective gatekeeper, allowing nutrients through and keeping everything else out. When it becomes more permeable than it should be (often called ‘leaky gut’), incompletely digested proteins can cross into the bloodstream, creating immune responses that contribute to a loss of self-tolerance over time.[6] Think of it like a nightclub with a bouncer who has stopped checking ID, all sorts of things get in that shouldn't, the wrong guests cause trouble and the clean-up takes considerably longer than the party.
Regulatory T cells are essentially your immune system's internal peacekeeping force, the calmer, quieter counterpart to the cells that mount the attack. Their job is to dampen the response when the threat has passed, to keep the immune system from overworking. They depend heavily on microbiome diversity (your friendly gut bugs) and the products (short-chain fatty acids) your gut bacteria produce, particularly butyrate. Butyrate supports the development of regulatory T cells by activating a transcription factor (FoxP3), which is crucial to their stability and function. When microbiome diversity is low, and in many modern Western guts it is considerably lower than it should be, that peacekeeping force can become underresourced.
Vitamin D directly influences the behaviour of the immune system's key decision-makers (dendritic cells), the ones that effectively tell other immune cells what is and isn't a threat. Vitamin D encourages dendritic cells to behave in a more tolerant, measured way, suppresses inflammatory pathways and supports the expansion of regulatory T cells (the peacekeepers). Deficiency is consistently associated with increased autoimmune risk.[7] Given that most of us in the UK are walking around measurably low in vitamin D for at least half the year, this is something worth considering. Explore more about the importance of vitamin D here.
Chronic stress activates the nervous system in ways that prime the immune system towards a more reactive, hypervigilant state, shifting it away from the measured, tolerant mode it needs for calm, appropriate responses. The stressed immune system is essentially running on high alert all the time, reacting first and asking questions later. Which, if you've been living with chronic symptoms alongside a full and demanding life, might sound rather familiar.
None of these are obscure or exotic factors. They are the fundamentals of autoimmunity and they are where the most meaningful work tends to happen. If you're interested in the specific role that viruses like Epstein-Barr (the glandular fever virus) play in this picture, I've written about its connection to multiple sclerosis here. You can also read more about how blood sugar and inflammation weave into the autoimmune story.
What this means for you
Functional medicine and nutritional therapy's role in autoimmunity is not to try to outsmart the immune system or to suppress it into submission. It's to understand what the immune system is trying to achieve, and then either support its ability to do that or remove the obstacles that are getting in the way.
That means asking different questions. Not just “what supplement or medication do I take for this condition?” but “what is driving the immune confusion and inflammation in this particular person, right now?" What needs to be removed? What needs to be restored? What does this person's timeline actually tell us about when things started to change and why?
Evidence increasingly suggests that the rise in autoimmune disease involves potentially modifiable risk factors and that socioeconomic, seasonal and regional variation points to environmental influences at play, not just genetics.[1] That is really significant as it means this is not simply a matter of the cards you were dealt, or ‘bad luck’ and it gives us the levers for potential change.
In summary, if you've skipped to the end
Autoimmunity is a loss of immune tolerance to the body's own proteins, leading to an inflammatory attack on specific tissues. The particular tissue being attacked determines the name of the condition. The immune system is not malfunctioning; it has been confused by earlier encounters… molecular similarities, gut permeability issues, environmental exposures, viruses and other factors that eroded its ability to properly distinguish itself from foreign invaders. Autoimmune disease typically develops over years before diagnosis, which means there is far more going on beneath the surface than a single test result or appointment can usually capture.
The same diagnosis presents differently in different people because the conditions that created it are unique to each individual. Those conditions: gut health, the microbiome, the nervous system, nutrient status, toxic load, hormonal balance, are exactly the territory that root-cause healing approaches work in.
The good news is that this is not a story about a broken, malfunctioning immune system. It's a story about a confused one, and confusion, with the right investigation, support and retraining can be addressed.
Ready to understand what's actually driving your symptoms?
If you've been handed a diagnosis, or you're still waiting for one, and you're ready to ask deeper questions, I'd love to talk.
The Get Empowered programme is built around exactly this kind of investigation: working through the root causes specific to you, rather than applying a one-size-fits-all approach to a highly individual process. Every case is unique and gets my undivided individual attention.
Has this resonated?
If you’ve been doing everything you can think of and still feel like something fundamental is missing, I would love to have a conversation with you. In my work with women navigating complex immune and chronic illness, reconnecting with the fundamentals - food, nature, nervous system regulation - is often where the most meaningful changes can happen.
If you would like to explore what that might look like for you specifically, book a free call here.
Make sure you are signed up to my Newsletter to receive my latest articles, recipes, recommendations and discounts to any upcoming events.
References
Conrad, N. et al. (2023) ‘Incidence, prevalence, and co-occurrence of autoimmune disorders over time and by age, sex, and socioeconomic status: a population-based cohort study of 22 million individuals in the UK’, The Lancet
Cojocaru, M. et al. (2011) ‘Pathogenesis of immune complexes in autoimmune diseases’, Maedica
Anaya, J.M. et al. (2018) ‘Molecular Mimicry and Autoimmunity’. Journal of Autoimmunity.
Eriksson, C. et al. (2011) ‘Autoantibodies predate the onset of systemic lupus erythematosus in northern Sweden’, Arthritis Research & Therapy.
van Delft, M.A.M & Huizinga T.W.J. (2020) ‘An Overview of Autoantibodies in Rheumatoid Arthritis’, Journal of Autoimmunity.;
and Arbuckle, M.R. et al. (2003) ‘Development of Autoantibodies Before the Clinical Onset of Systemic Lupus Erythematosus’, New England Journal of Medicine.
Fasano, A. (2020) ‘All Disease Begins in the (Leaky) Gut: role of zonulin-mediated gut permeability in the pathogenesis of some chronic inflammatory diseases’, F1000Research.
Hahn, J. et al. (2022) ‘Vitamin D and marine omega 3 fatty acid supplementation and incident autoimmune disease: VITAL randomized controlled trial’, BMJ.